People with allergy-induced asthma dread the time of year when pollen blankets cars, sidewalks, and anything outside. Even a gentle breeze causes individuals with the condition to experience such symptoms as wheezing, airway constriction, and lung inflammation.
Thanks to work performed by researchers at the National Institutes of Health (NIH), people with allergic asthma may be closer to having new therapies. The research was published April 1 in the Journal of Clinical Investigation.
 “My group is interested in different forms of asthma, including allergic asthma, which is characterized by the accumulation of eosinophils,” Cook said. (Photo courtesy of Steve McCaw / NIEHS)
“My group is interested in different forms of asthma, including allergic asthma, which is characterized by the accumulation of eosinophils,” Cook said. (Photo courtesy of Steve McCaw / NIEHS)Scientists at NIEHS and the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) found a new molecular pathway that worsens allergic asthma in mice and possibly humans. The pathway involves three components:
- A cell surface receptor called P2Y14.
- A sugar known as uridine diphosphate glucose (UDP-G).
- Eosinophils, which are specialized white blood cells (see sidebar).
Understanding the pathway
According to Donald Cook, Ph.D., head of the NIEHS Immunogenetics Group and corresponding author of the study, asthma has two phases. The first phase, called the sensitization phase, is similar to what happens after a person receives a vaccination against a viral or bacterial infection.
'The first time a person is exposed to an allergen, he or she can become immunized against it, just like a person can become immunized to a virus after receiving a vaccine,' Cook said.
Immune cells remember what the allergen looks like and can respond when they see it again, he explained. However, repeated exposures will trigger immune responses that lead to airway inflammation and other features of asthma. In mouse models of asthma, these immune responses are the second phase, or the challenge phase.
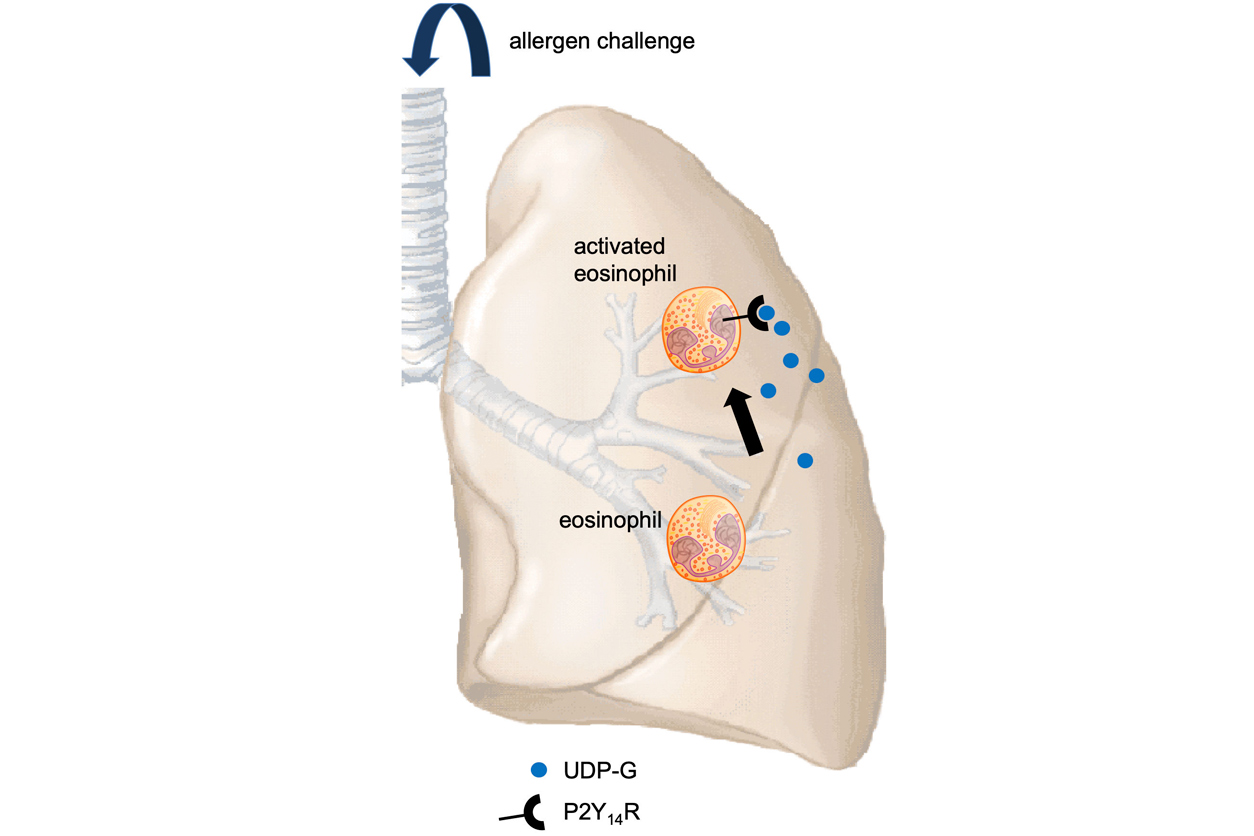 During allergen challenge, eosinophils travel to the lung, contributing to shortness of breath. This is driven partly by UDP-G production and interaction with the P2Y14 receptor. Antagonists that block this interaction reduce eosinophils. (Image courtesy of Donald Cook / NIEHS)
During allergen challenge, eosinophils travel to the lung, contributing to shortness of breath. This is driven partly by UDP-G production and interaction with the P2Y14 receptor. Antagonists that block this interaction reduce eosinophils. (Image courtesy of Donald Cook / NIEHS)Cook said that UDP-G is present in mice airways normally, but its levels increase substantially during the challenge phase. This is when UDP-G binds to the P2Y14 receptor and promotes eosinophilic inflammation and airway constriction.
Cook theorized that the P2Y14/UDP-G pathway promotes eosinophil migration to the lung, which is consistent with a 2017 genome-wide association study, or GWAS, that showed P2Y14 may be involved in human asthma.
Therapeutic compounds
To test the therapeutic potential of the P2Y14/UDP-G pathway, Cook and his colleagues gave asthma model mice P2Y14 compounds that bind to P2Y14, but do not activate it like UDP-G. These are called antagonists. When an antagonist binds to P2Y14, it prevents UDP-G from binding.
One of those compounds, called PPTN, is commercially available. Experiments showed that PPTN reduced eosinophilic inflammation in the mouse asthma models. The findings suggest it may have similar effects in human asthma, representing a potential therapy.
 “Chemistry within the [NIH] Intramural Research Program has an important role in the discovery of new disease treatments,” Jacobson said. (Photo courtesy of NIDDK)
“Chemistry within the [NIH] Intramural Research Program has an important role in the discovery of new disease treatments,” Jacobson said. (Photo courtesy of NIDDK)'We discover and chemically synthesize new drugs in our lab,' said Kenneth Jacobson, Ph.D., head of the Molecular Recognition Section in the NIDDK Laboratory of Bioorganic Chemistry. 'Our focus on P2Y and other related receptors has been fruitful in the search for clinical candidate molecules, such as potent and selective P2Y14 antagonists.'
NIEHS-NIDDK partnership
Jacobson has been working with the P2Y14 receptor for many years and reached out to Cook to join forces on this project. Jacobson also provided novel, high affinity antagonists that are being tested in the same mouse model of asthma. Cook and Jacobson anticipate that these compounds, or their derivatives, could one day be used to reduce the severity of allergic asthma in humans.
Their collaboration was possible because several years ago, NIEHS Scientific Director Darryl Zeldin, M.D., and his counterpart, NIDDK Scientific Director Michael Krause, Ph.D., decided to fund cooperative ventures between the two institutes. This research is an excellent example of what can happen when two NIH institutes work together.
'The joint NIEHS-NIDDK fellowship program is now in its sixth year and has really stimulated productive scientific interactions between investigators in the two institutes,' Zeldin said.
Krause agreed. 'It is gratifying to see that this program is fostering collaborations that are producing outstanding science, realizing the main goal we envisioned for this institute partnership from the start,' he said.
Citations:
Karcz TP, Whitehead GS, Nakano K, Nakano H, Grimm SA, Williams JG, Deterding LJ, Jacobson KA, Cook DN. 2021. UDP-glucose and P2Y14 receptor amplify allergen-induced airway eosinophilia. J Clin Invest 131(7):e140709.
Ferreira MA, Jansen R, Willemsen G, Penninx B, Bain LM, Vicente CT, Revez JA, Matheson MC, Hui J, Tung JY, Baltic S, Le Souef P, Montgomery GW, Martin NG, Robertson CF, James A, Thompson PJ, Boomsma DI, Hopper JL, Hinds DA, Werder RB, Phipps S, Australian Asthma Genetics Consortium Collaborators. 2017. Gene-based analysis of regulatory variants identifies 4 putative novel asthma risk genes related to nucleotide synthesis and signaling. J Allergy Clin Immunol 139(4):1148–1157.