In addition to allowing humans and animals to breathe, lungs also play a role in the immune response and are heavily affected by external conditions. The lungs are constantly exposed to the environment and are susceptible to inflammatory diseases, like asthma, which are influenced by substances in the air.
 Garantziotis said the results confirmed the research group’s hypothesis. (Photo courtesy of Steve McCaw)
Garantziotis said the results confirmed the research group’s hypothesis. (Photo courtesy of Steve McCaw)In two newly published papers, a team led by NIEHS scientists described how environmental exposures promote and worsen asthma. The mechanism involves the immune pathway of a protein called toll-like receptor 5 (TLR5). More than 26 million Americans have asthma, so the findings may have important implications for easing the severity of the disease.
Companion papers
Stavros Garantziotis, M.D., medical director of the NIEHS Clinical Research Unit (CRU) and head of the Matrix Biology Group, is corresponding author of both articles. He said the first paper, published in Elife Jan. 28, described the TLR5 mechanism. Researchers used an NIEHS-funded study group of healthy individuals (see top sidebar) who were exposed to ozone concentrations similar to an Air Quality Index alert level of orange or red.
Participants’ lungs responded by producing airway inflammation, but the study found individuals who lacked a functional TLR5 receptor had much less lung inflammation.
Team members found a similar result when blood cells from participants in the NIEHS Environmental Polymorphisms Registry (EPR), a North Carolina DNA bank used to find environmental determinants of human disease, were exposed to a common bacterial component called endotoxin.
The second paper, published in Lung on Feb. 14, went a step further and concluded that the activator of the TLR5 pathway is found not only in pollutants, but also in homes. The researchers examined EPR participants with asthma rather than healthy individuals. They noticed that those with a nonfunctional TLR5 experienced less asthma symptoms. The team also confirmed in a mouse study that activating TLR5 promoted worsening of asthma.
 'The papers show that TLR5 not only supports detection of endotoxin by TLR4, but the findings also have a much broader role in physiology as a detector of tissue damage elicited by noninfectious environmental stress,' Fessler said. (Photo courtesy of Steve McCaw)
'The papers show that TLR5 not only supports detection of endotoxin by TLR4, but the findings also have a much broader role in physiology as a detector of tissue damage elicited by noninfectious environmental stress,' Fessler said. (Photo courtesy of Steve McCaw)'We’re in a situation where we can really understand the mechanisms that lead to human disease like asthma,' Garantziotis said. 'We now know what causes its activation, what it leads to, and how we can intervene to make it less active.'
NIEHS groups working together
Michael Fessler, M.D., chief of the NIEHS Immunity, Inflammation, and Disease Laboratory worked with Garantziotis on the Elife paper. Donald Cook, Ph.D., head of the Immunogenetics Group, did a lot of work for the Lung article (see sidebar).
The EPR was instrumental in both papers. The registry contains health and exposure information for thousands of participants.
NIEHS Clinical Director, Janet Hall, M.D., recognized its research significance for NIEHS. 'This resource is poised to play an increasingly important scientific role as we seek to understand the ways in which genes and environment interact in human health,' Hall said.
Environmental precision medicine
Garantziotis said taken together, the papers have two implications. The first is personalized medicine. Scientists can ask which exposures in a person’s house and environment can make a person sicker and what is it about that person’s biology makes them more or less susceptible. In the case of a nonfunctional TLR5, the person is less susceptible to asthma. By default, if a person has a functional receptor, they are more susceptible to those exposures.
 'These new findings extend previous work by showing that the flagellin component of house dust likely contributes to asthma flare ups, known as exacerbations,' Cook said. (Photo courtesy of Steve McCaw)
'These new findings extend previous work by showing that the flagellin component of house dust likely contributes to asthma flare ups, known as exacerbations,' Cook said. (Photo courtesy of Steve McCaw)Second, the results may suggest environmental mitigation steps. If a TLR5 activator exists in someone’s home, it makes them more susceptible to asthma. If the activator is removed, their asthma may become less active. It is a genetic perspective that translates into positive health outcomes in real life.
Citations:
Hussain S, Johnson CG, Sciurba J, Meng X, Stober VP, Liu C, Cyphert-Daily JM, Bulek K, Qian W, Solis A, Sakamachi Y, Trempus CS, Aloor JJ, Gowdy KM, Foster WM, Hollingsworth JW, Tighe RM, Li X, Fessler MB, Garantziotis S. 2020. TLR5 participates in the TLR4 receptor complex and promotes MyD88-dependent signaling in environmental lung injury. Elife; doi: 10.7554/eLife.50458 [Online 28 January 2020].
Whitehead GS, Hussain S, Fannin R, Trempus CS, Innes CL, Schurman SH, Cook DN, Garantziotis S. 2020. TLR5 activation exacerbates airway inflammation in asthma. Lung; doi: 10.1007/s00408-020-00337-2 [Online 14 February 2020].
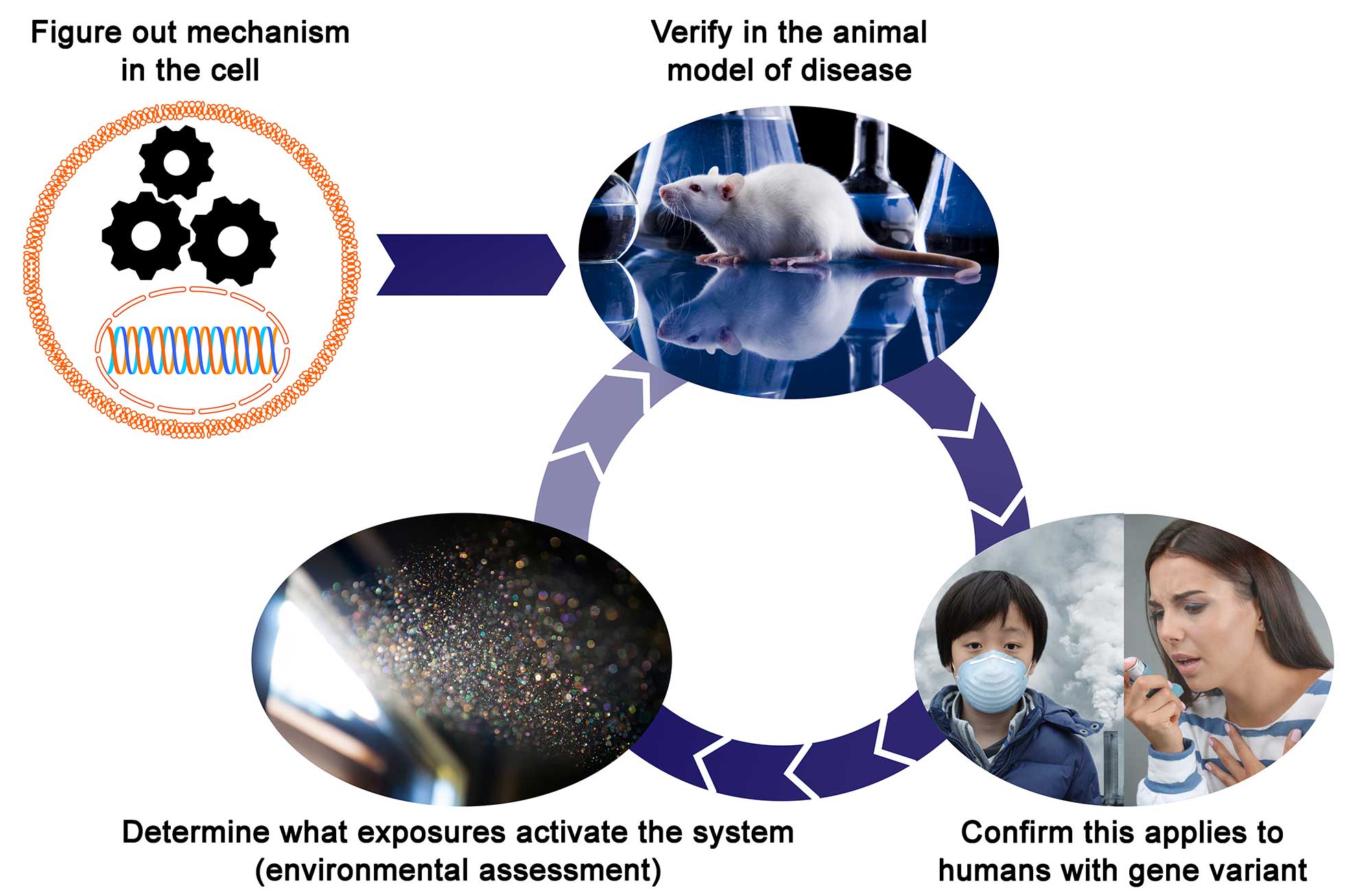
The graphic illustrates how the two papers fit together. First, the investigators figured out the mechanism of TLR5 signaling, shown by gears and DNA in a cell. Next, they worked out the implications in an animal model and then in humans after exposure to ozone and then endotoxin. Then they changed the study scenario to house dust as the exposure asthma patients as the population. This process shows how genes and environment together affect human disease.