NIEHS researchers have developed a new tool called a systematic evidence map that enables more informed decision-making regarding the links between personal care products and early puberty. The findings were published in the November issue of Environment International.
“Researchers and regulators can use the map to identify gaps in data and existing scientific studies, which will help target and prioritize future research and funding,” said lead author Kyla Taylor, Ph.D., a health scientist in the Division of Translational Toxicology (DTT) at NIEHS.
The map provides a foundation to inform and direct new approaches for evaluating evidence about exposure to chemicals in personal care products and puberty-related health effects.

Efficiently evaluating endocrine-disrupting chemicals
Personal care products (PCPs), as the name suggests, are consumer products used for personal hygiene, personal grooming, or beautification. They contain many different ingredients and can be a source of exposure to endocrine-disrupting chemicals (EDCs), including phthalates and phenols.
EDCs are substances or mixtures of substances that interfere with normal hormone function. Early-life exposure to EDCs commonly found in PCPs has been linked to earlier onset of puberty.
To address public health concerns regarding environmental exposures, researchers have conducted hazard and risk assessments that evaluate scientific evidence, prioritize research needs, and inform regulatory decisions. But these assessments are typically done on a chemical-by-chemical basis and require significant time and resources. There is a growing need for a more efficient approach, particularly given the increasing number of chemicals in use.
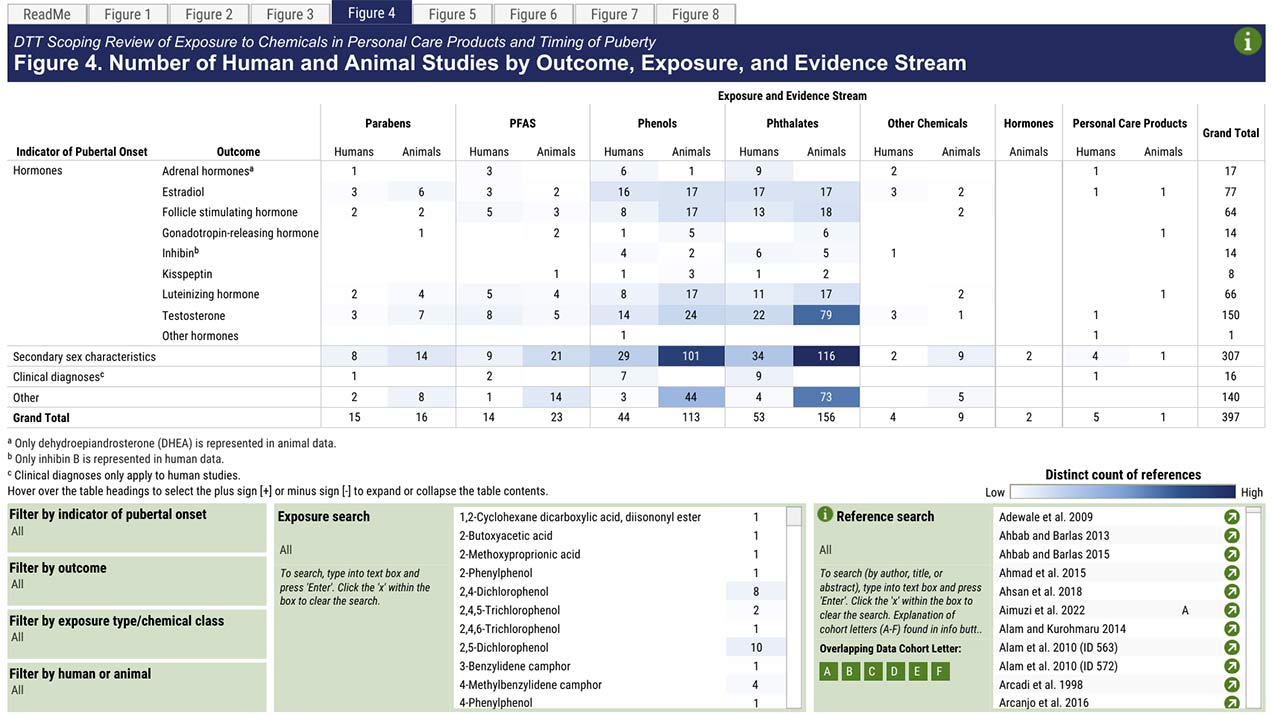
Grouping chemicals into categories or classes can enable more efficient decision-making. These class-based approaches group multiple chemicals together in relation to physiochemical, environmental, and toxicological properties. In the new study, the researchers set out to inform decision-making on a class-based assessment of PCPs and puberty.
“Prior to this study, there had been a lack of comprehensive, systematic reviews that outlined the human and animal evidence of a link between EDCs found in PCPs and timing of puberty,” said senior author Andrew Rooney, Ph.D., acting chief of the Integrative Health Assessments Branch.

Interactive visualizations
The researchers analyzed 98 human studies and 299 animal studies that evaluated a total of 96 different chemicals. Phthalates and phenols were the most well-studied chemical classes, and most of these studies examined secondary sex characteristics, such as breast development and changes in levels of estradiol and testosterone hormones. Only three studies included multiple racial or ethnic categories and reported results by these categories, and none analyzed results according to socioeconomic status (see sidebar).
Based on the results, the researchers developed an interactive visualization that supports evidence-based decisions on the available research. The tool enables people to search, sort, and filter the existing scientific literature on puberty-related studies by key concepts, including the type of PCP, health outcome, study design, and health disparities.
“Because we classified individual chemicals into broad chemical classes, this systematic evidence map can be used to identify the chemical classes with sufficient puberty-related health outcome data, which will inform a systematic review for a hazard or risk assessment,” said Taylor.
The researchers believe their findings support the development of a focused systematic review to evaluate and integrate the human and animal evidence of the association between specific puberty-related health outcomes and phthalates and phenols found in PCPs. Such a review is critical for reaching hazard conclusions based on the totality of the evidence, and clearly communicating potential harmful effects from chemicals in personal care products to the public.
(Janelle Weaver, Ph.D., is a contract writer for the NIEHS Office of Communications and Public Liaison.)









