Epigenetics, which refers to the study of heritable changes in gene expression that do not involve alterations in DNA sequence, offer a glimpse into changes that may lead to development of cancers, according to Yvonne Fondufe-Mittendorf, Ph.D., from the University of Kentucky. In a Nov. 27 talk for the NIEHS Keystone Science Lecture Seminar Series, Fondufe-Mittendorf shared her passion for discovering processes that explain the scientific observations made in her lab.
 Fondufe-Mittendorf is more than a grantee — she also makes important contributions to environmental health science through service, according to Tyson. (Photo courtesy of Steve McCaw)
Fondufe-Mittendorf is more than a grantee — she also makes important contributions to environmental health science through service, according to Tyson. (Photo courtesy of Steve McCaw)Interactions between the epigenome and environmental insults, particularly at critical stages of development, may cause cancers and other diseases, she noted. Cell-based research in her lab identified key molecular players and shed considerable light on the mechanistic pathways involved (see citations below).
Joining chromatin biology with toxicology
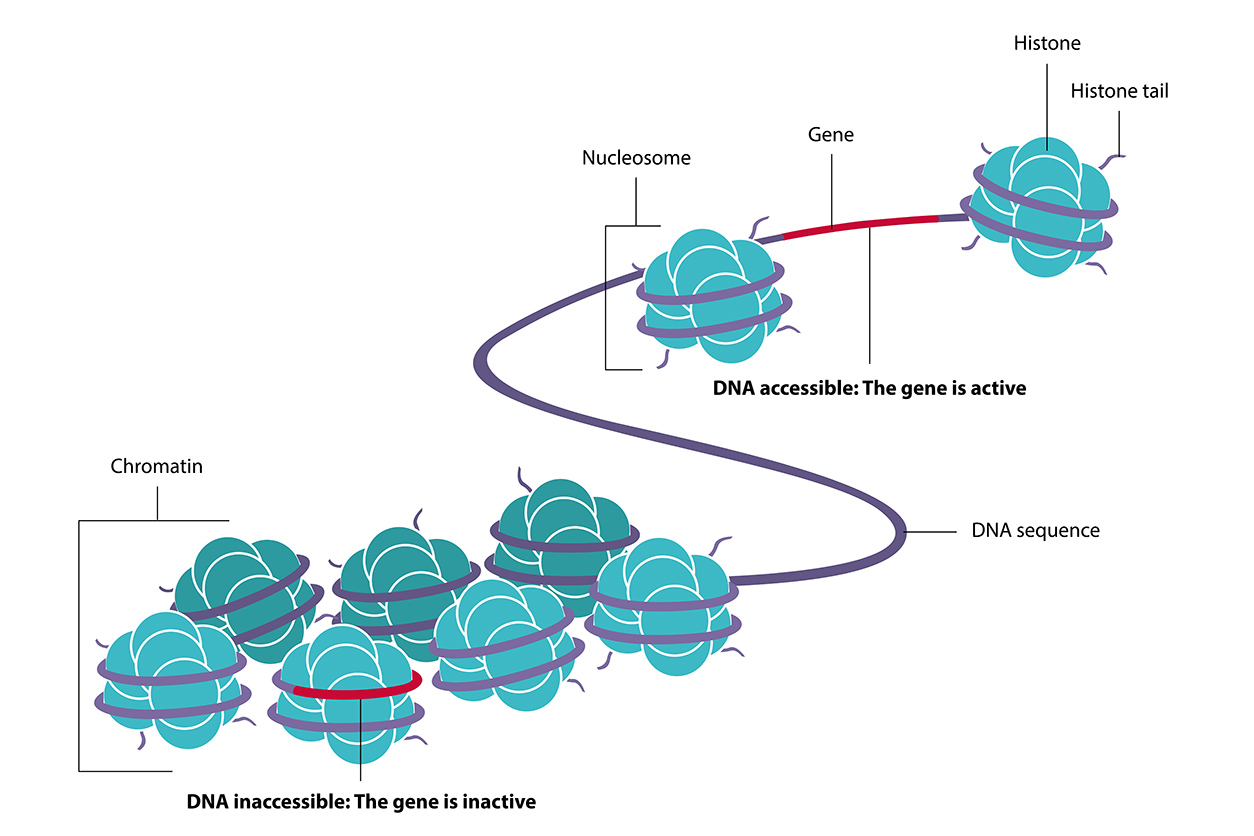
The Fondufe-Mittendorf lab focuses on chromatin, and she describes herself as a chromatin biologist who has added toxicology to her work. Chromatin is the complex formed by DNA and histones, which are proteins that package DNA in the nucleus of a cell.
Chromatin modifications are an important kind of epigenetic change because they can affect how gene transcription and splicing are regulated, she said. Such changes are sensitive to environmental cues, including exposure to inorganic arsenic, a commonly occurring metal that affects gene regulation through epigenetic changes.
“Dr. Fondufe-Mittendorf’s research captured my attention several years ago when she applied to the TaRGET I [grant program],” said NIEHS Program Director Fred Tyson, Ph.D., who hosted the talk. “Since that time, she has begun exploring mechanisms that explain how arsenic can alter transcriptional regulation by disrupting the three-dimensional [3D] architecture of chromatin. This is fascinating work.”
Real-world impact
Eastern Kentucky is coal mining country, so Fondufe-Mittendorf’s work extends far beyond the lab bench. Exposure to inorganic arsenic has been linked to lung cancer and other health concerns.
“I see a great need in the Appalachian region, because residents are exposed to contaminants that seep into drinking water due to coal mining activities,” she said, noting that unfiltered well water is a commonly used source of drinking water.
Arsenic, zinc, and cancer
 Tyson introduced Fondufe-Mittendorf, and moderated the question and answer session following her presentation. (Photo courtesy of Steve McCaw)
Tyson introduced Fondufe-Mittendorf, and moderated the question and answer session following her presentation. (Photo courtesy of Steve McCaw)Fondufe-Mittendorf has shown that long-term, low-dose exposure to inorganic arsenic can transform normal cells into carcinogenic cells. “We now have a model to understand the molecular underpinnings as cells go through carcinogenesis,” she said.
Tyson pointed to her studies (see citations below) showing that arsenic can replace zinc in certain sites on regulatory proteins. “This can be associated with abnormal chromatin looping, which can affect transcription, and may be involved with development of cancer,” he said.
Fondufe-Mittendorf reported that inorganic arsenic affects the binding of a protein called CTCF, especially at sites where CTCF binds weakly. CTCF is referred to as one of the master weavers of the genome, she explained, because it helps to wire the genome into a 3D shape.
The study of this shaping has been limited, but new techniques allow researchers to learn more about how it is formed, regulated, and dysregulated in diseases such as cancer. Fondufe-Mittendorf said her research is helping deepen understanding of how changes in CTCF binding affect the rewiring of the genome. Such knowledge is critical to developing therapeutics.
She plans to investigate public health measures to combat the cancer-causing effects of arsenic exposure, including the possibility of administering zinc. “It is possible that just by administering zinc supplements, we might lessen the effects of inorganic arsenic’s displacement of zinc in regulatory proteins such as CTCF,” Fondufe-Mittendorf said. “This is noninvasive and a very simple way that we can actually help people treat the problems [arsenic] is associated with.”
 The room was filled with engaged attendees, many of whom lined up afterward to speak with Fondufe-Mittendorf about her research (Photo courtesy of Steve McCaw)
The room was filled with engaged attendees, many of whom lined up afterward to speak with Fondufe-Mittendorf about her research (Photo courtesy of Steve McCaw)A bigger picture
The idea that cancer may derive from epigenetic changes rather than just genetic mutations may have profound implications well beyond the Appalachian region and exposures to inorganic arsenic.
“The arsenic work was an amazing case study, a proof of principle that will no doubt spawn entirely new lines of research,” said Tyson. “That’s why I wanted to bring Dr. Fondufe-Mittendorf here. It’s exciting research.”
Citations:
Rea M, Gripshover T, Fondufe-Mittendorf Y. 2018. Selective inhibition of CTCF binding by iAs directs TET-mediated reprogramming of 5-hydroxymethylation patterns in iAs-transformed cells. Toxicol Appl Pharmacol 338:124–133.
Eckstein M, Rea M, Fondufe-Mittendorf YN. 2017. Transient and permanent changes in DNA methylation patterns in inorganic arsenic-mediated epithelial-to-mesenchymal transition. Toxicol Appl Pharmacol 331:6–17.
Eckstein M, Eleazer R, Rea M, Fondufe-Mittendorf Y. 2017. Epigenomic reprogramming in inorganic arsenic-mediated gene expression patterns during carcinogenesis. Rev Environ Health 32(1–2):93–103.
Rea M, Eckstein M, Eleazer R, Smith C, Fondufe-Mittendorf YN. 2017. Genome-wide DNA methylation reprogramming in response to inorganic arsenic links inhibition of CTCF binding, DNMT expression and cellular transformation. Sci Rep 7:41474.
Riedmann C, Ma Y, Melikishvili M, Godfrey SG, Zhang Z, Chen KC, Rouchka EC, Fondufe-Mittendorf YN. 2015. Inorganic arsenic-induced cellular transformation is coupled with genome wide changes in chromatin structure, transcriptome and splicing patterns. BMC Genomics 16:212.
(Ernie Hood is a contract writer for the NIEHS Office of Communications and Public Liaison.)